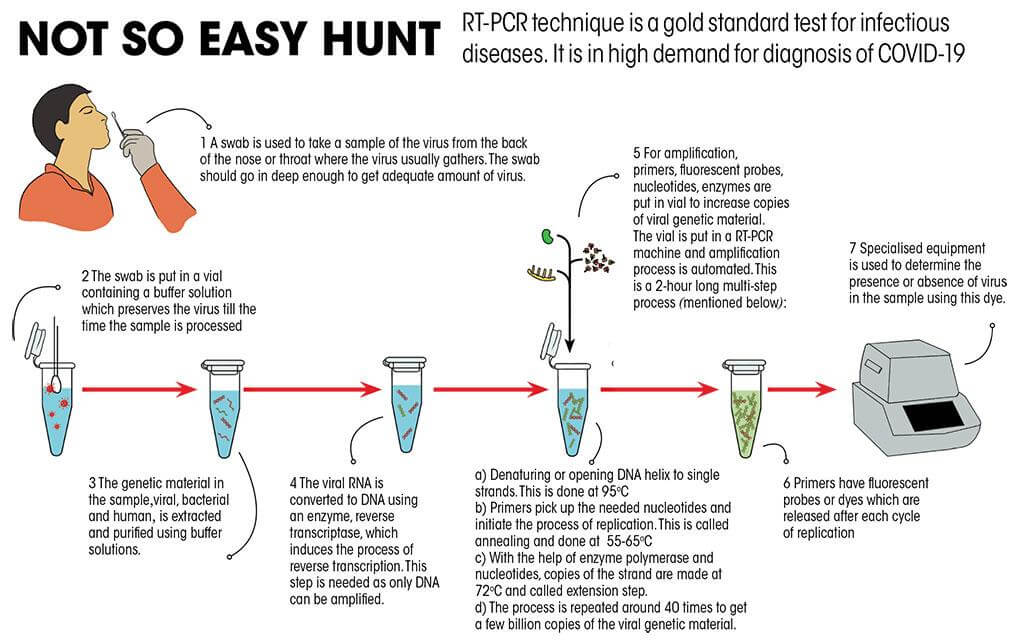
designed to detect only one viral target on the SARS-CoV-2 genome.Tests with "single target" in the attribute column are: In addition to COVID-19 tests issued EUAs, there are molecular SARS-CoV-2 diagnostic tests that have been authorized through traditional premarket review pathways. To see additional authorization documents, such as letters granting EUA amendments or revisions, and a list of other brand names authorized under a specific EUA, select the plus (+) button beside the "Date EUA Issued or Last Updated" for the EUA.įor information on test EUAs that have been revoked see Historical Information about Device Emergency Use Authorizations: In Vitro Diagnostics (IVD). Tests available without a prescription include the attribute "DTC" (for direct-to-consumer home collection tests) or "OTC" (for over-the-counter at-home tests). For example, tests authorized for the screening of asymptomatic individuals without known exposure are listed with "screening" in the attribute column pooling, multi-analyte, saliva, home collection, and home testing are similarly listed. Test attributes are listed in the "Attributes" column. These emergency use authorizations (EUAs) have been issued for each individual test with certain conditions of authorization required of the manufacturer and authorized laboratories. The table below includes information about authorized SARS-CoV-2 molecular diagnostic tests. Individual EUAs for Molecular Diagnostic Tests for SARS-CoV-2 EUA for Molecular Diagnostic Tests for SARS-CoV-2 Developed And Performed By Laboratories Certified Under CLIA To Perform High Complexity Tests.Umbrella EUA for SARS-CoV-2 Molecular Diagnostic Tests for Serial Testing.Pooling and Serial Testing Amendment for Certain Molecular Diagnostic Tests for SARS-CoV-2.Individual EUAs for Molecular Diagnostic Tests for SARS-CoV-2.IVDs for Management of COVID-19 Patients.Serology and Other Adaptive Immune Response Tests for SARS-CoV-2.Antigen Diagnostic Tests for SARS-CoV-2.In Vitro Diagnostic EUAs: Overview and Templates Webinar on Guidances on COVID-19 Transition Plans for Medical Devices - April 18, 2023.Transition Plan for Medical Devices Issued Emergency Use Authorizations (EUAs) Related to Coronavirus Disease 2019 (COVID-19). 
Transition Plan for Medical Devices That Fall Within Enforcement Policies Issued During the Coronavirus Disease 2019 (COVID-19) Public Health Emergency.In particular, for manufacturers planning to seek marketing authorization for their devices, the FDA recommends beginning work on a marketing submission, including a transition implementation plan, as described in the guidances. The FDA encourages stakeholders to review the two final guidances, view the webinar, and reach out to the FDA if they have questions or concerns. Taking other actions with respect to these devices.Developing a transition implementation plan,.The guidances outline the FDA's general recommendations to transition from certain policies adopted and operations implemented during the COVID-19 pandemic to normal operations, including the FDA's recommendations for: MaThe FDA has finalized two guidances: Transition Plan for Medical Devices That Fall Within Enforcement Policies Issued During the Coronavirus Disease 2019 (COVID-19) Public Health Emergency and Transition Plan for Medical Devices Issued Emergency Use Authorizations (EUAs) Related to Coronavirus Disease 2019 (COVID-19).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
